Lewis dot structures YouTube
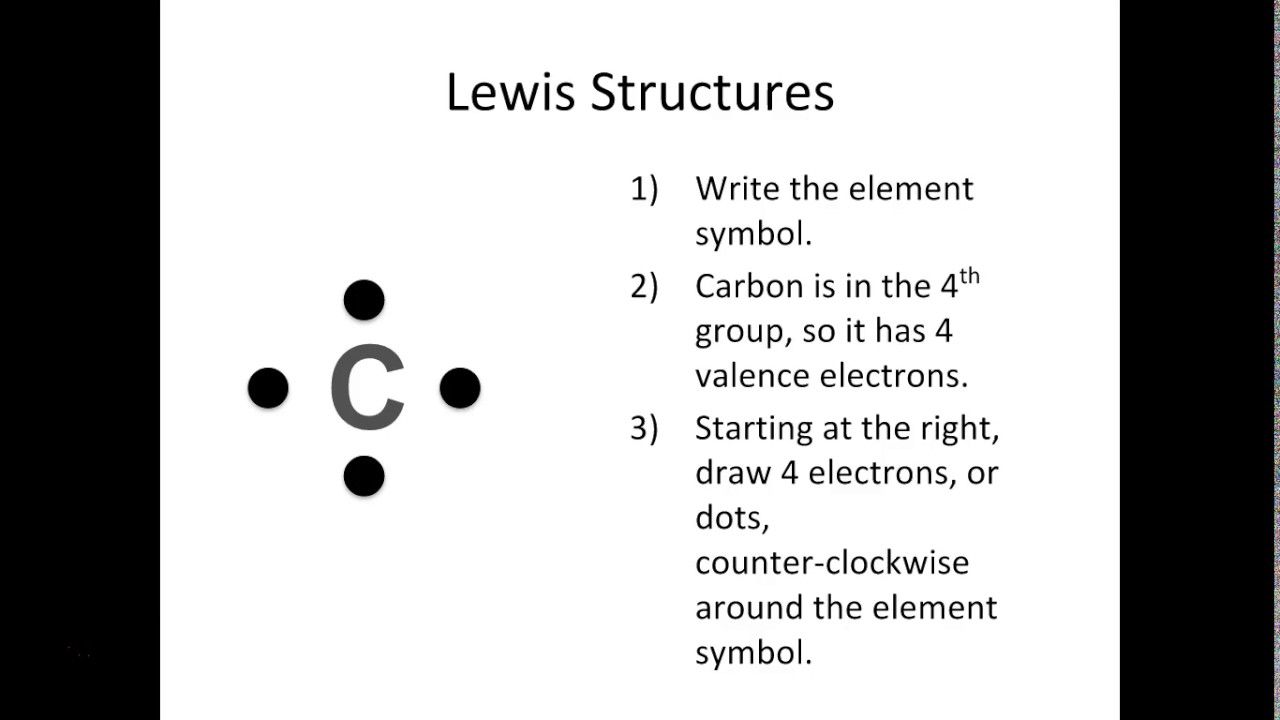
Follow these simple steps to draw Lewis dot structures: Draw the atoms on paper and put dots around them to represent valence electrons of the atom. Be sure to have the correct number of electrons. If the species is an ion, add or subtract electrons corresponding to the charge of the ion.
Lewis dot structures of atoms and ions pogil answers
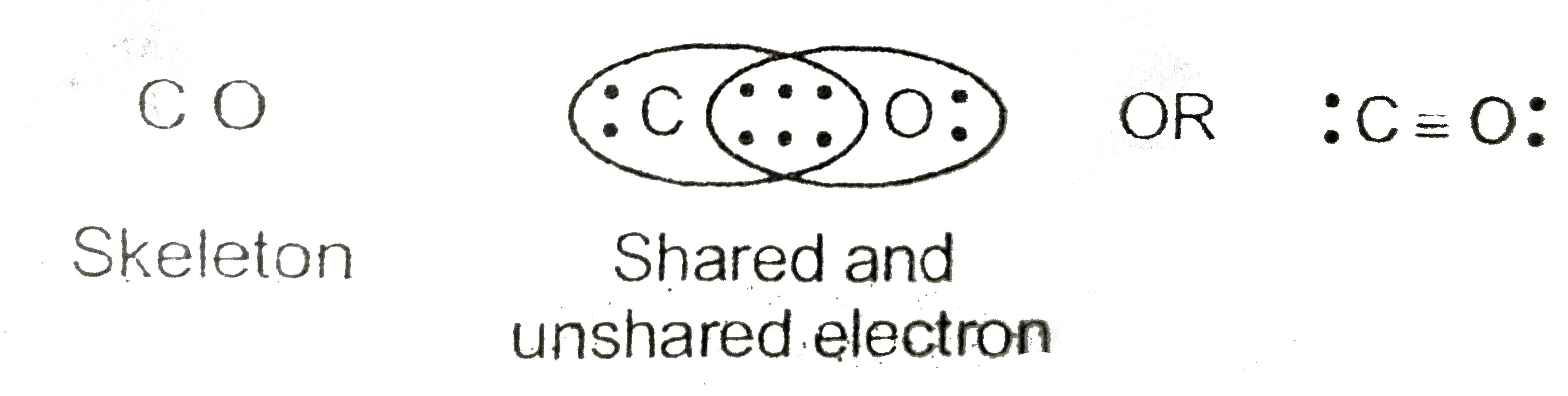
That is CO. Carbon has 4 valence electrons and Oxygen has 6. In total, there are 10 valence electrons. I realize that explaining how to draw the lewis dot structure in words might get confusing so what I'm going to do is put a picture of what the diagram will look like along with a video showing step by step on how to exactly create the diagram.

CO Lewis Structure How to Draw the Dot Structure for CO YouTube
The Lewis Dot Structure (Lewis Dot Diagram) of or for CO.1.Count valence electrons of CO2.Keep the least electronegative atom in centre3.Put one valence e.

CO (Carbon Monoxide) Lewis Dot Structure with Formal Charge
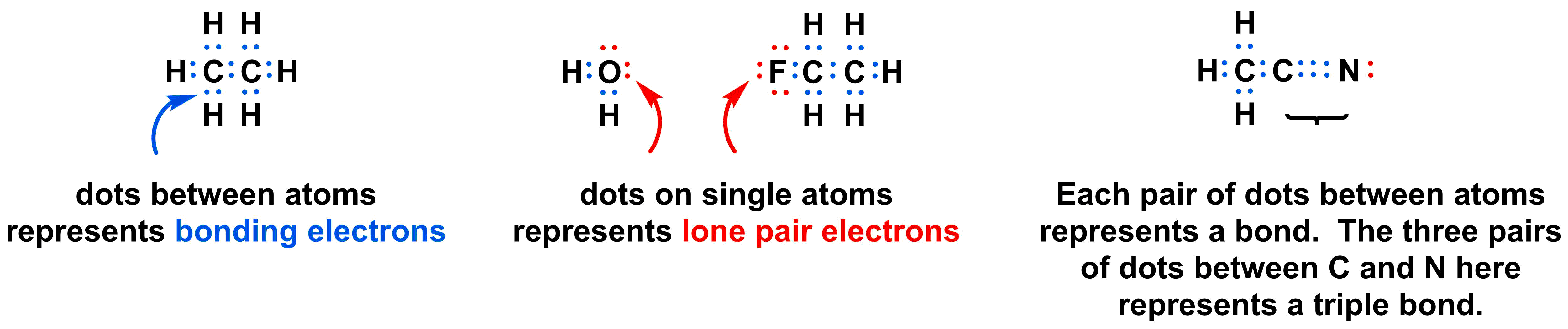
Lewis structures (also known as Lewis dot diagrams, electron dot diagrams,"Lewis Dot formula" Lewis dot structures, and electron dot structures) are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. A Lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. The Lewis structure.

Lewis Dot Diagram For F Drivenheisenberg
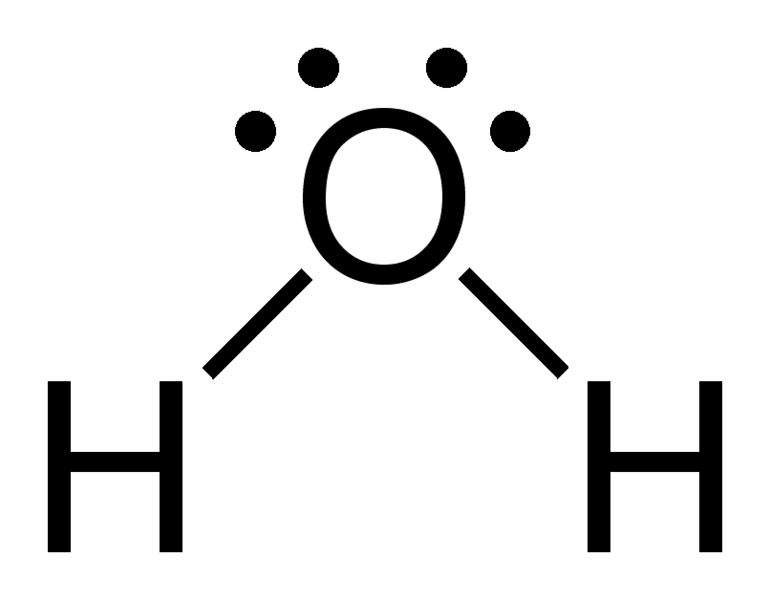
Solution. Solutions to Example 10.4.1. Steps for Writing Lewis Structures. Example 10.5.1 10.5. 1. 1. Determine the total number of valence electrons in the molecule or ion. Each H atom (group 1) has 1 valence electron, and the O atom (group 16) has 6 valence electrons, for a total of 8 valence electrons.
9.1 Chemical Bonds, Lewis Symbols, and the Octet Rule Chemistry
The Lewis structure, also called as electron dot structure, is a simplified method of representing the number of valence electrons present within an atom or a molecule. Furthermore, the structure helps with determining the number of lone pairs of electrons present in an atom and how they act in a bond formation.
Carbon Monoxide Dot Structure
Lewis dot structures also called electron dot structures are diagrams that describe the chemical bonding between atoms in a molecule. They also display the total number of lone pairs present in each of the atoms that constitute the molecule. Lewis dot structures are commonly referred to as electron dot structures or Lewis structures.

CO (Carbon Monoxide) Lewis Dot Structure with Formal Charge
The Lewis Structure (Lewis Dot Diagram) for CO. 1. Count electrons 2. Put least electronegative atom in centre.more.more The Lewis Structure (Lewis Dot Diagram) for CO.1..

Write the Lewis dot structure of CO molecule.
Chemistry 101A Topic F: Molecular Structure 9: Basic Concepts of Covalent Bonding 9.3: Drawing Lewis Structures
14+ Co Lewis Dot Structure Robhosking Diagram
A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 7.9 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 7.9 Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table.
Lewis Dot Structure Generator demorejaz
A step-by-step explanation of how to draw the CO Lewis Dot Structure (Carbon monoxide ).For the CO structure use the periodic table to find the total number.

Lewis Dot Diagram Worksheet
Lewis Dot Structure of CO (Carbon Monoxide) kentchemistry.com 25.1K subscribers Subscribe Subscribed Share 151K views 12 years ago Every Video I quickly take you through how to draw the.
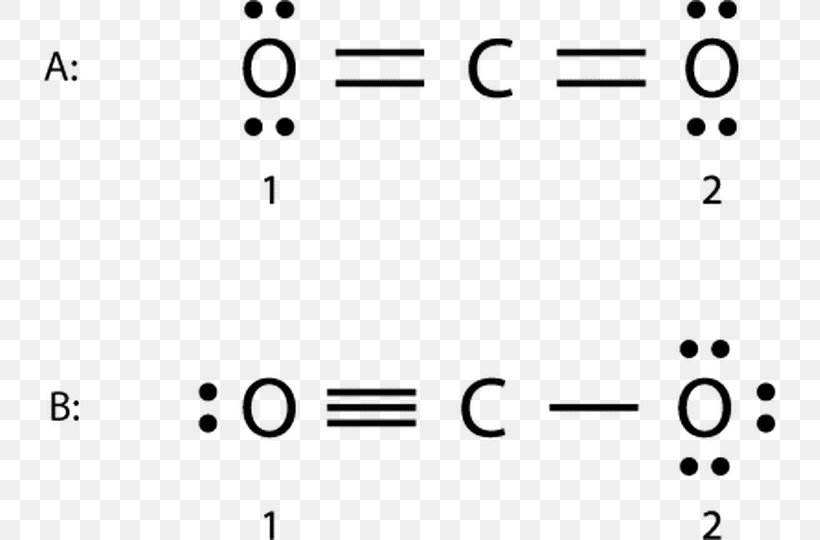
Formal Charge Lewis Structure Resonance Chemistry Carbon Dioxide, PNG
How to Draw the Lewis Dot Diagram for Carbon monoxide (CO) It is helpful if you: Try to draw the CO Lewis structure before watching the video. Watch the video and see if you missed any steps or information. Try structures similar to CO for more practice. List of Lewis Structures Lewis Structures for CO.
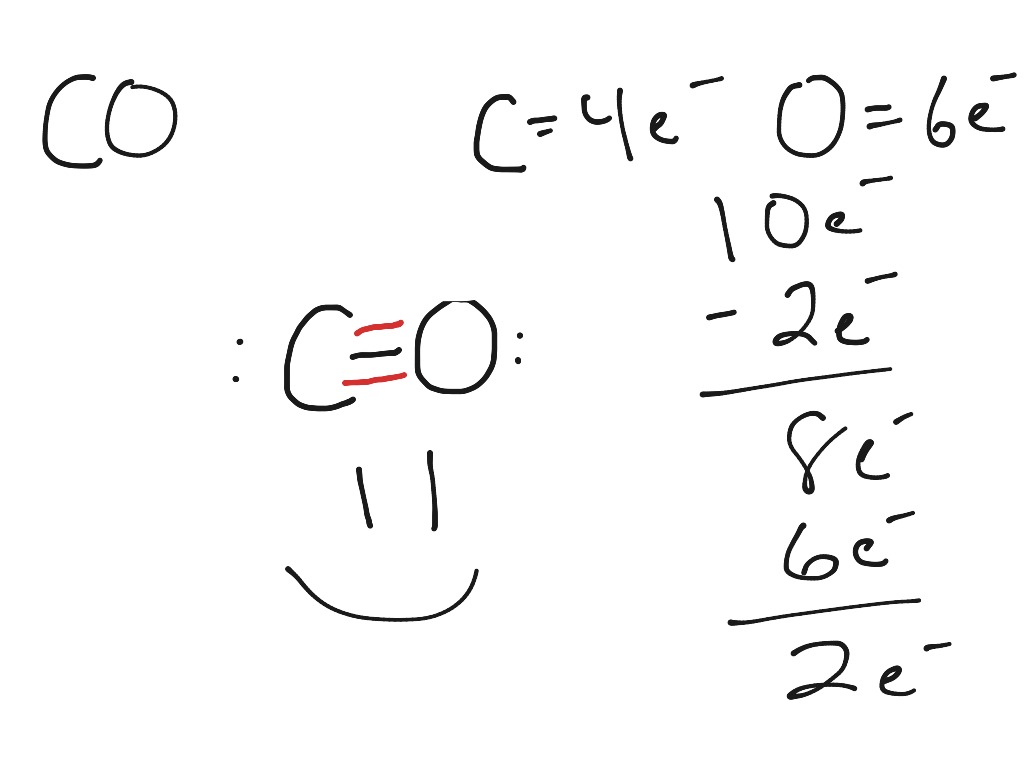
Electron Dot Diagram For Carbon General Wiring Diagram
A step-by-step explanation of how to draw the CO Lewis Dot Structure (Carbon dioxide). For the CO structure use the periodic table to find the total number of valence electrons for t.more.
Laura September 2010
CO. Back. 70 More Lewis Dot Structures. Produced from the incomplete combustion of hydrocarbons. In the winter, it is important that furnaces have access to an ample supply of oxygen in the air. It is a colorless, odorless, tasteless gas that highly toxic to humans. CO contains a covalent double bond and a third bond that is considered to be.

How to Draw Lewis Dot Structure
The N atom has the following Lewis electron dot diagram: It has three unpaired electrons, each of which can make a covalent bond by sharing electrons with an H atom. The electron dot diagram of NH 3 is as follows: Exercise 12.4.2 12.4. 2. Use a Lewis electron dot diagram to show the covalent bonding in PCl 3. Answer.